Sensitivity range: 0.3 – 5.0 µm
This product is obsolete effective December 31, 2022.
Simplifies air particle monitoring for remote locations.
As part of a facility monitoring solution, the IsoAir® 310P Particle Sensor from Particle Measuring Systems (PMS) makes it easy to monitor air in remote locations or in areas with process restraints.
With the successful introduction of the new IsoAir® Pro-E and IsoAir Pro-Plus Aerosol Particle Sensors, we are announcing the obsolescence of the IsoAir® 310P. The last day that Particle Measuring Systems will accept orders for this model is April 15, 2022.
Real-time particle monitoring gives quick notification of particle anomalies, detecting 0.5 and 5.0 µm for GMP and FDA compliance. A central vacuum is not needed to operate the particle sensor, and it features a built in “low noise” blower together with a Hepa filtered exhaust.
It has unparalleled performance in a chemical-resistant and easy-to-disinfect 316 L box, and the passivated stainless steel surface has well defined microbial control characteristics.
The clean, reliable internal pump lowers installation costs and the need for costly vacuum system connections; the instrument is available with a wide range of communications protocol such as Ethernet, Modbus, Wifi, Analog 4-20mA for full compatibility with the existing monitoring system.
Features
- Four standard particle channel sizes with two-channel pharmaceutical mode
- Flow rate of 1.0 CFM
- Passivated 316L stainless-steel enclosure
- Automatic shutoff for capped probe, flow sensing
- Compliance with ISO 21501-4 and CE standards
- Two local LEDs for status and activity, easy to see and understand status lights
- Optional 4-20 mA inputs allow for integration of up to three environmental input sensors
- Queues 3,000 cleanroom samples in the event of computer system failure
- Available validation documentation
Benefit
- Ethernet, Modbus and 4-20 mA outputs meet 21 CFR Part 11 regulations for data transmission security
- Identifies breakdown in isolator protection
- Interfaces with optional FacilityPro, Pharmaceutical Net Pro or FacilityNet software for comprehensive management of your cleanroom
- Automatic paging allows rapid response to alarms
- Easy to remove particle sensor simplifies calibration and service
- Diode laser reduces maintenance requirements
- Pump with HEPA filtered exhaust protects from creation of more particles
Applications
- Pharmaceutical manufacturing area
- Isolator monitoring
- Remote cleanroom monitoring
- Aerospace and industrial applications
- Trend analysis
- Statistical process control
Supporting Materials
Webinars
Tackling the 21CFR11 Challenge: From Paper to Paperless >
Viable Non Viable Particle Counts to Fully Meet Regulatory Requirements >
Steps to Ensuring a Successful Audit – Effective Risk Assessment Design Webinar >
Private: EU GMP Annex 1 Review, Insights, and Feedback >
Assuring Data integrity in an Environmental Monitoring System >
Available Accessories
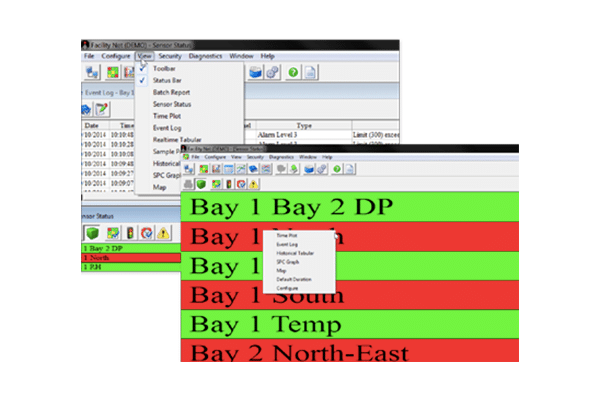
Facility Net
Facility Net Software provides a solution for comprehensive environmental monitoring, including a real-time and historical data display. Compatible with Particle Measuring Systems particle counters and molecular contamination monitors, as well as various third-party products.
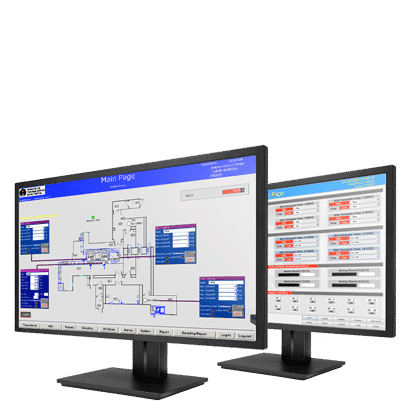
FacilityPro® Software
FacilityPro® Software provides the interface, data management, and reporting for an environmental monitoring system. FacilityPro’s ability to manage viable, nonviable and environmental data through a common system improves the efficiency of production operations and quality investigations.

Differential Air Pressure Sensor
The Differential Air Pressure Sensor allows for accurate measurement of air with a range of 0 to 0.25in (0 to 6.35mm) of water. Current output: 4-20mA.