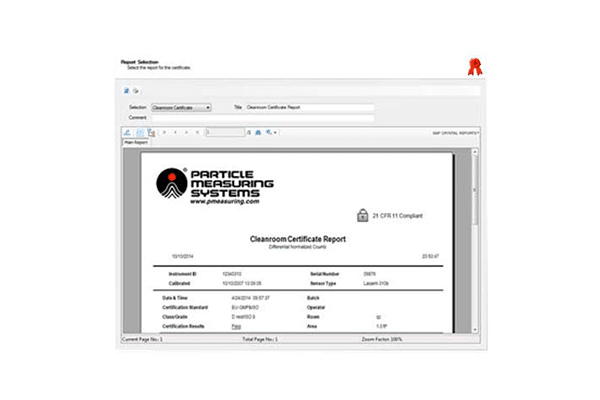
21 CFR Part 11 Compliant
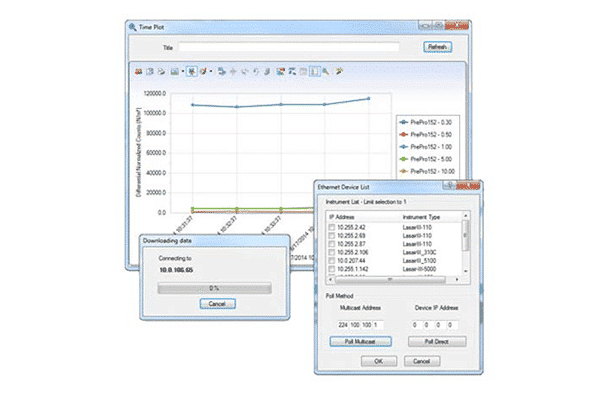
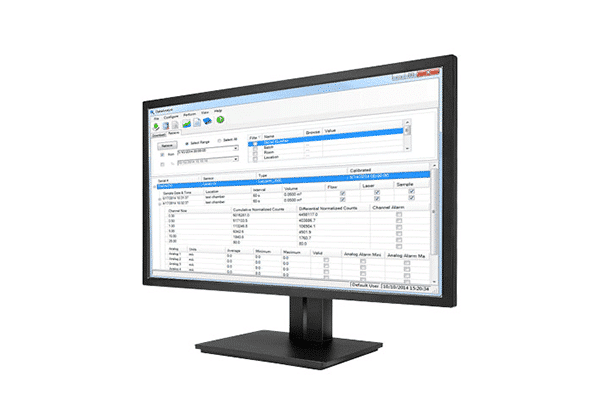
DataAnalyst Particle Counter Software by Particle Measuring Systems PMS helps you understand the data coming from your particle monitor for an easy and low-cost way to effectively manage data, including maintaining electronic records. DataAnalyst Particle Counter Software knows what to look for and does the calculations for you. It’s easy to download, archive, and report data that is fully compliant with all secure data transfer requirements.
The comprehensive reporting structure works with specific inputs from the particle counter and filters sampling data based on location, date/time, or product batch information. Reports can be generated to meet industry requirements and configured to include raw data, tabular data, and statistical and graphical formats. The reporting tool can also be configured to give various report formats, including cleanroom certification to ISO14644, FDA 21 CFR Part 11, and EU GMP Annex 1.
The integration with the Lasair® Pro Particle Counter or MiniCapt® Mobile microbial samplers allows for the collection, storage, and reporting of sampling data. The new manual data entry function enables the authorized user to include CFU counts, incubation temperature, and incubation time.
All cleanroom classification and monitoring data is now in a unique, secure, and validated place.
Product Details
Resources
Educational Material
- Compressed Gases: An Important Component of an Environmental Monitoring Program in a Pharmaceutical Manufacturing Environment
- Data Integrity: Understand and Comply with GMP and FDA Requirements for 21 CFR Part 11
- The Data Quality Concept – A New Challenge for Quality Experts
Not sure where to start? Here are the next steps:

Tell us about your application requirements.

Our experts will help you find the right solutions to meet your specific requirements.

Once we identify the best solution for you, we provide you with pricing and delivery dates.